Neon is a much more common element in stars and is the fifth most abundant element in the universe. It can be produced commercially from liquid air through a process called fractional distillation. 
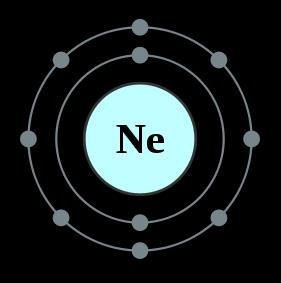
It is found in very small traces in both the Earth's atmosphere and the Earth's crust. When neon is in a vacuum discharge tube, it glows with a reddish-orange light. It is the second lightest noble gas after helium. It only remains a liquid from 24.55 K to 27.05 K. Neon has the narrowest liquid range of any element. It is a completely inert gas, meaning that it will not combine with other elements or substances to create a compound. Under standard conditions the element neon is a colorless odorless gas. Neon atoms have 10 electrons and 10 protons with a full outer shell of 8 electrons. Neon is the fifth most abundant element in the universe. Neon is the second noble gas located in column 18 of the period table. Discovered by: Sir William Ramsay and M.Throughout this scientific discovery and analysis process of all three naturally-occurring neon isotopes, it is now being theorized that neon can be useful in determining cosmic exposure ages of meteorites and rocks located on a planetary body’s surface. Due to the enriched amounts of 20Ne in volcanic gas and its presence in diamonds, some theorize that there may exist a “solar neon reservoir” in the Earth. The relative sources of 20Ne, while the most abundant of the three isotopes on earth, is debated to this day. 20Ne, on the other hand, is known to be primordial but not nucleogenic. The nucleogenic instances of neon isotopes are known to be a result of nuclear reactions, primarily with 24Mg and 25Mg. 21Ne and 22Ne are both primordial (have existed since the formation of the earth) and nucleogenic (produced through naturally-occurring nuclear reactions). Neon has three stable isotopes: 20Ne, 21Ne, and 22Ne. It has 40 times the refrigerant capacity of liquid helium and three times the refrigerant capacity of liquid hydrogen. 
Lesser known applications include neon as a refrigerant in its liquid form. For this reason, neon was the precursor of today’s modern plasma displays and TV screens. Neon itself is reddish-orange when activated, but lighting apparatuses can replicate a number of colors in the visible spectrum when neon is mixed with other gases. Its attractive characteristics in lighting applications are enabled by its activation (atom separation and recombination in a vacuum tube) at nominal voltages and currents in modern electrical systems. Because of these two factors, neon can be a relatively expensive element to produce and purchase. Neon is an iconic element and can only be produced through fractional distillation of liquefied air. This method of isolating atmospheric elements was clearly fruitful as Ramsay won the Nobel Prize for Chemistry in 1904 for these numerous discoveries. In fact, this rare atmospheric element was discovered a mere two weeks after another noble gas, krypton. Neon was discovered, as with many noble gases, through fractional distillation of liquefied air in 1898 by British chemists Sir William Ramsay and Morris Travers. It is theorized that neon’s volatile nature with high vapor pressure and relative lightness caused the element to escape in large quantities during the formation of Earth as a planet, under the sun’s blanket of heat. No known neutral compounds exist with this chemically unreactive element, and therefore neon cannot affix itself to any solids. Neon is a noble gas, and perhaps surprisingly, the fifth most plentiful element in the cosmos (after hydrogen, helium, oxygen and carbon) however, its abundance in Earth’s atmosphere is rather rare (about 18 ppm). American Elements: The Materials Science Company™ | Certified bulk & lab quantity manufacturer of metals, chemicals, nanoparticles & other advanced materials 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
